In the Earth's atmosphere we find short-lived and longer-lived gases.
The phenomena of air quality and climate are linked to atmospheric gases and the lifetime of a molecule. The latter is an indication of the time it takes for a molecule to be converted into another substance via chemical reactions in the atmosphere.
Sunlight very often plays an important role here. Many chemical reactions in the atmosphere take place under the influence of the sun.
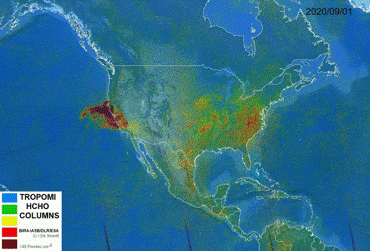
Short-lived gases (seconds, hours) = air quality
If a substance has a short lifetime (from a few seconds to a few hours), it does not have the chance to transport itself far from its emission source.
This means that we observe short-lived gases close to their emission source (e.g. industry plant, forest fire, …) allowing us to detect emission hotspots very easily.
Examples of gases with high relevance to air quality:
- Ammonia - NH3
- Formaldehyde - HCHO
- Nitrogen dioxide - NO2
- O3 at ground level
- Sulphur dioxide - SO2
- Glyoxal - CHOCHO
- PM - Particulate Matter - Aerosols (particles in suspension, liquid or solid)
Longer-lived gases (weeks, months, years) = climate
The longer-lived gases (weeks, months, years) have more time to be transported over long distances (up to hundreds of kilometers) and possibly to higher altitudes before they chemically react with other substances in the atmosphere.
This means that they are much more mixed in the atmosphere. Therefore, if we observe such a long-living gas somewhere, we cannot simply say that it was emitted in the immediate vicinity. It is possible that it was transported to the observation site from longer distances.
Examples of gases with high relevance to climate:
- Carbon dioxide - CO2
- Methane - CH4
- Water vapor - H2O
- Chlorofluorocarbons - CFCs
- Sulfur hexafluoride - SF6
- Hydrogen chlorofluorocarbons - HCFCs
- Tropospheric ozone - O3
Air quality and climate linked through chemical reactions
In general, the short-lived gases are primarily relevant to air quality and the longer-lived gases more relevant to climate, but there is a very strong link between the two phenomena.
Many of them affect both air quality and climate.
This is because one gas is converted into another through a variety of complex chemical reactions. Often the end product of a whole complex chemical chain, is a series of short-lived gases, which eventually are converted into long-lived gases.
Examples of complex atmospheric chemistry:
- More intense solar radiation causes the production of more ozone smog in the air and the release of more volatile hydrocarbons by plants and trees. This in turn affects the formation of dust and tropospheric ozone. With increased heat episodes due to global warming, humans will seek more cooling, thus consuming more energy and releasing more greenhouse gases and air pollutants into the atmosphere.
- All kinds of particles can reflect sunlight back to a greater or lesser extent, causing cooling or warming. Soot, for example, absorbs radiation. When soot particles land on a layer of snow, less solar radiation bounces back, contributing to global warming. Sulphur particles bounce back radiation and have a cooling effect.
Reducing emissions of short-lived climate pollutants
It may be of interest to policy makers to pay particular attention to reducing emissions of molecules that both pollute the air and have a greenhouse effect, the so-called "short lived climate pollutants". These are e.g.
- methane
- ozone
- soot (black carbon)
- Halogenated hydrofluorocarbons (HCFCs).
Due to the relatively short lifespan of many of these substances, measures have a rapid effect. Addressing these substances will help both improve air quality and reduce the rate of climate change.
Watch more:
Video animation explaining the connections between air pollution and climate change
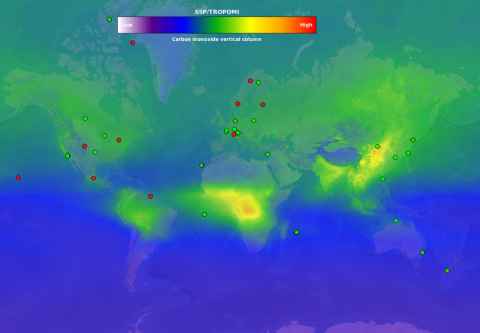
CO observations are used to study both climate change and air quality in the atmosphere. Tropospheric CO is created by incomplete combustion (of fuels, biomass) and by oxidation of methane and other hydrocarbons.