Origin, properties and effects of NO2
NO2 or nitrogen dioxide is a substance released by combustion processes.
Over three-quarters (78%) of the air we breathe is composed of nitrogen (N2). At high temperatures, nitrogen reacts with oxygen (O2) to form nitrogen monoxide (NO) and nitrogen dioxide NO2. NO is further converted to NO2 in the atmosphere. The collective name for NO and NO2 is nitrogen oxides (NOx).
N2 + O2 → NO (→ NO2) and partly NO2
NO2 is a reactive gas ("strong oxidant") involved in the formation of ozone and particulate matter (smog) and is known to be irritating to the human respiratory system. It has a significant effect on human health and contributes to respiratory problems:
- irritation nose, throat, eyes, lungs
- asthma attacks
- respiratory complaints
Furthermore, when an excess of nitrogen compounds is released into the atmosphere, these can wreak havoc on many earth and water-based ecosystems. They cause the acidification of the soil, thus interfering with the growth of plants in the wild or in agriculture. When they are dissolved in water, they impact the chemical processes and balance, which can lead to health issues and even death of marine and freshwater life.
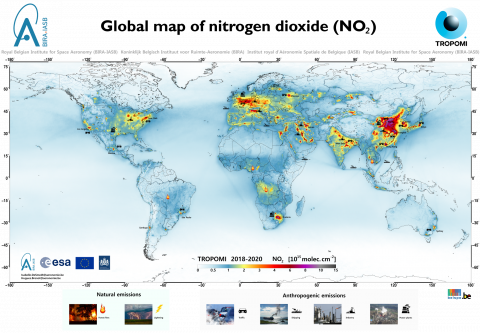
NO2 sources from human activity (65% of total nitrogen dioxide emissions)
Nitrogen dioxide in the atmosphere has both natural and anthropogenic sources, with humans causing the vast majority (65%), through emissions from:
- cars
- ships
- airplanes
- industrial processes
- power plants
The combustion of oil, coal and gas is responsible for increasing levels of carbon dioxide (CO2) in the atmosphere, one of the main causes of global warming. In addition to large amounts of NO2 and CO2, those combustion processes of heating systems, power generation and engines (transportation) also release other pollutants into the atmosphere, such as:
- sulphur dioxide (SO2)
- formaldehyde (HCHO)
Despite increased awareness around the carbon footprint and the impact of energy consumption, the use of non-renewable fossil fuels continues to increase worldwide for the time being.
Natural sources of NO2 (35% of total nitrogen dioxide emissions)
Nitrogen dioxide is also released from natural combustion processes such as forest fires and lightning. In addition, plants and bacteria can also produce NO2 in the soil or water.